What Is The Function Of Schwann Cells – The presence of bones around the central nervous system (CNS) provides it with highly effective physiologically crucial mechanical protection. The peripheral nervous system (PNS), on the other hand, lacks this barrier. Consequently, the long-held belief is that the PNS is mechanically vulnerable. On the other hand, the PNS is exposed to a variety of physiological mechanical stresses during regular daily activities. This fact leads us to question the dogma of PNS mechanical vulnerability. Indeed, impaired mechanics of PNS nerves is associated with neuropathies with propensity to mechanical loads in parallel with significant impairment of PNS physiological functions. Our recent biomechanical integrity studies on nerve fibers from wild-type and neuropathic mice provide strong support in favor of natural mechanical protection of the PNS and demonstrate a key role for Schwann cells (SCs) therein. Moreover, recent works point out that SCs can sense mechanical properties of their microenvironment, and evidence is growing that SCs mechanosensitivity is important for PNS development and myelination. Therefore, SCs exhibit mechanical strength necessary for PNS mechanoprotection as well as mechanosensitivity necessary for PNS development and myelination. This mini-review reflects on the intriguing dual capability of SCs and implications for PNS physiology and pathophysiology.
The general assumption that the peripheral nervous system (PNS) is prone to mechanical damage is based on the absence of a formidable physical barrier in the PNS that presents the skull and bony vertebrae in the central nervous system (CNS). However, lack of mechanical protection or mechanical strength should inhibit the physiological activities of the PNS. Take, for example, the primary nerve function that is the propagation of action potentials. Conduction velocity is used as a criterion to classify nerves. While some nerves exhibit a conduction velocity as low as 0.1 ms
What Is The Function Of Schwann Cells

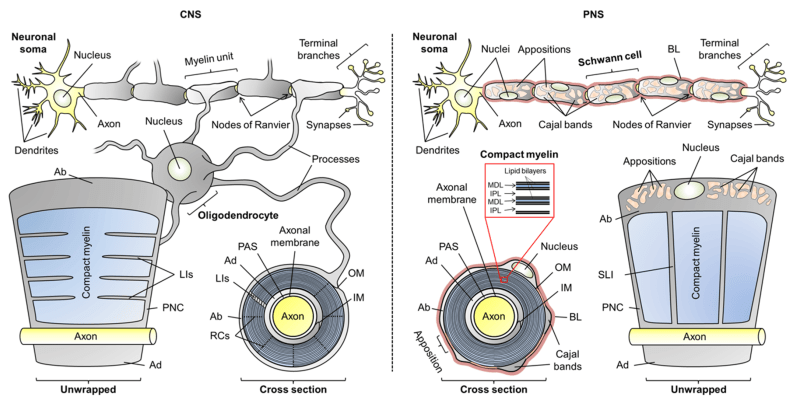
Which is of crucial importance for vital physiological reactions (Debanne, 2004). The presence of a myelin sheath around nerve fibers is one of the critical factors that ensure a high conduction velocity. Another critical factor is axon caliber; the bigger the faster. Consequently, proneness to mechanical compression would significantly slow conduction velocity or induce conduction block, and the resulting pathophysiological consequences would be severe. Therefore, the assumption that PNS lacks mechanical strength is somewhat shaky and should be reconsidered. The question is relevant as to whether the main components of PNS provide the natural mechanical strength. Likely candidates are several tissues and extracellular matrices (ECMs) within PNS nerves (Colognato et al., 2005), which may offer some mechanical support. In the PNS, each individual nerve fiber is wrapped in a protective sheath known as the endoneurium. In addition, fibers are bundled into fascicles with a protective lining called the perineurium. Fascicles are again connected by a blood supply and fatty tissue in yet another layer known as the epineurium. Unlike oligodendrocytes and their associated axons in the CNS, Schwann cells (SCs) and axons in the PNS are enveloped in a basal lamina, a specialized form of ECM (Colognato et al., 2005). The rather thin basal lamina is mainly secreted by SCs (Obremski et al., 1993). Various roles have been proposed for the basal lamina, including SC proliferation, survival, migration and myelination (Colognato et al., 2005; Court et al., 2006; Chernousov et al., 2008; Colognato and Tzvetanova, 2011; Pereira et al. al., 2012). The role of the basal lamina in providing mechanical strength to PNS nerves has recently been demonstrated ( Rosso et al., 2014 ) and will be discussed in this review. Moreover, recent observations in vitro show that SCs are able to sense the mechanical stiffness of their ECM protein-coated substrate (Rosso et al., 2017). Mechanosensitivity greatly affects SCs shape, migration, adhesion and mechanics (Rosso et al., 2017) and is therefore assumed to play key roles in PNS development, maintenance, myelination and regeneration as discussed here.
Role Of Connexin Based Gap Junction Channels In Communication Of Myelin Sheath In Schwann Cells
It should be noted that the interactions of cells with ECMs are not only biochemical in nature, but also highly mechanical (Discher et al., 2005). There is overwhelming evidence for the importance of mechanical interactions between ECM and cells as shown for different cell types (Even-Ram et al., 2006; Discher et al., 2009; Franze, 2013). Tissue cells are naturally not suspended in liquid, but anchored, which is essential for their viability. Anchorage, on the other hand, exposes cells to mechanical forces imposed on them by the physical properties of their environment, particularly the ECM stiffness (Discher et al., 2005). Almost two decades ago, a study was published that revealed how differently fibroblasts looked and moved on substrates with the same chemical properties but different stiffness (Pelham and Wang, 1997). Intriguingly, fibroblast movement and focal adhesion were regulated by the stiffness of their substrate. Inspired by these results, various follow-up studies were published (Engler et al., 2004, 2006; Gilbert et al., 2010; Arulmoli et al., 2015; Sosale et al., 2015). In some cases, the mechanical properties of the microenvironment were observed to modify or even override chemical signals (Engler et al., 2006). Therefore, in the meantime, it is without doubt that feedback from local ECM stiffness on cell state has profound consequences for development, differentiation, disease and regeneration (Discher et al., 2005). However, the exact mechanism underlying stiffness sensing remains unclear (Tee et al., 2009; Moore et al., 2010; Franze et al., 2013; Humphrey et al., 2014). Mechanosensing the environment is followed within microseconds by transmission of the gathered information to the cell nucleus to trigger a biochemical response in a physiological process known as mechanotransduction (Jaalouk and Lammerding, 2009; Wang et al., 2009). Cytoskeletal proteins, integrins, nesprins and nuclear lamina are among the various proteins involved in the transmission process (Jaalouk and Lammerding, 2009; Wang et al., 2009). Mutation or misregulation of proteins involved in mechanotransduction is closely associated with the onset and progression of various diseases ranging from muscular dystrophies to cardiomyopathies, cancer and neuropathies among others (Franze, 2013; Franze et al., 2013). Numerous studies highlight the importance of mechanotransduction for the nervous system, giving rise to a new branch of neuroscience called neuromechanics (Franze, 2011). Different types of neurons and glial cells in the CNS have been shown to respond strongly to the stiffness of their substrate (Flanagan et al., 2002; Georges et al., 2006; Jiang et al., 2008; Rosenberg et al., 2008; Moshayedi et al., 2010). Substrate stiffness has a major influence on the morphology, development, network, dynamics, behavior and function of neuronal and glial cells (Kostic et al., 2007; Previtera et al., 2010; Cellot et al., 2011; Fabbro et al., 2012; Tang et al., 2013; Zhang et al., 2014). Neuronal cells can even use stiffness gradients of their environment as a guide for their migration, a process referred to as mechanotaxis (Franze, 2011). Unlike the CNS, which has certainly received some attention so far, the importance of mechanosensitivity for the PNS is just beginning to be recognized (Koch et al., 2012; Athamneh et al., 2015; Poitelon et al., 2016; Urbanski et al., 2016; Rosso et al., 2017).
The main glial cells in the PNS are SCs. Generation of SCs is a multistep process called SC lineage (Mirsky and Jessen, 1996; Jessen and Mirsky, 2005). It starts from the migrating neural crest cells of the dorsal root ganglion, which first develop into SC precursors and then into immature SCs (Jessen and Mirsky, 2005). Immature SCs undergo a transition into two different cell types, myelinating and non-myelinating SCs. This decision by immature SCs about their fate as myelinating or nonmyelinating is still puzzling (Jessen and Mirsky, 2005). All immature SCs are considered to share the same developmental potential and their decision to become myelinating or non-myelinating is determined by the axon they connect to Jessen and Mirsky (2005). It is well known that only axons larger than ∼1 μm in diameter are myelinated ( Jessen and Mirsky, 2005 ) and that axonal Neuregulin-1 (NRG1) signaling is a prerequisite ( Michailov et al., 2004 ; ffrench-Constant et al. ., 2004; Taveggia et al., 2005; Pereira et al., 2012; Heller et al., 2014). Nevertheless, the triggers for myelination and the resulting downstream signaling are still a matter of intense debate (Pereira et al., 2012). The two types of SCs play different roles in the PNS and both are essential for its maintenance (Jessen and Mirsky, 1999). The production and wrapping of myelin around axons allows rapid conduction of signaling essential to nervous system function. In addition to myelin production, SCs perform several tasks that are indispensable for the development, physiological support, survival, protection, and regeneration of the PNS (Jessen and Mirsky, 1999). Several of these functions are in turn mediated by the basal lamina (Chernousov et al., 2008), whose production is a sequential process starting at the stage of immature SCs (Jessen and Mirsky, 2005).
Exposure of SCs to mechanical force begins right from the early embryonic stages and is maintained throughout life. Among the major forces is the significant increase in the stiffness of SCs’ microenvironment along their morphogenesis (Figure 1). This is due to progressive production of connective tissue and basal lamina, which peaks at the stage of mature SCs (Jessen and Mirsky, 2005; Figure 1). Finally,
What is the function of b cells, structure and function of schwann cells, what are schwann cells, what do schwann cells do, what is the function of mast cells, location of schwann cells, what is the purpose of schwann cells, what is the function of stem cells, what is the function of white cells, purpose of schwann cells, function of schwann cells, what is the function of t cells